What Is the Bayesian Statistical Approach?
The Bayesian approach is a method for making decisions under uncertainty by combining what you already know with new evidence. Named
after 18th-century mathematician Thomas Bayes, it provides a formal framework for updating beliefs as data accumulates.
Think of it like an experienced physician diagnosing a patient. A doctor doesn't start from scratch with each case, they bring years of knowledge about disease prevalence, symptom patterns, and treatment outcomes. When new test results arrive, they update their assessment accordingly. Bayesian statistics formalizes this intuitive process mathematically.
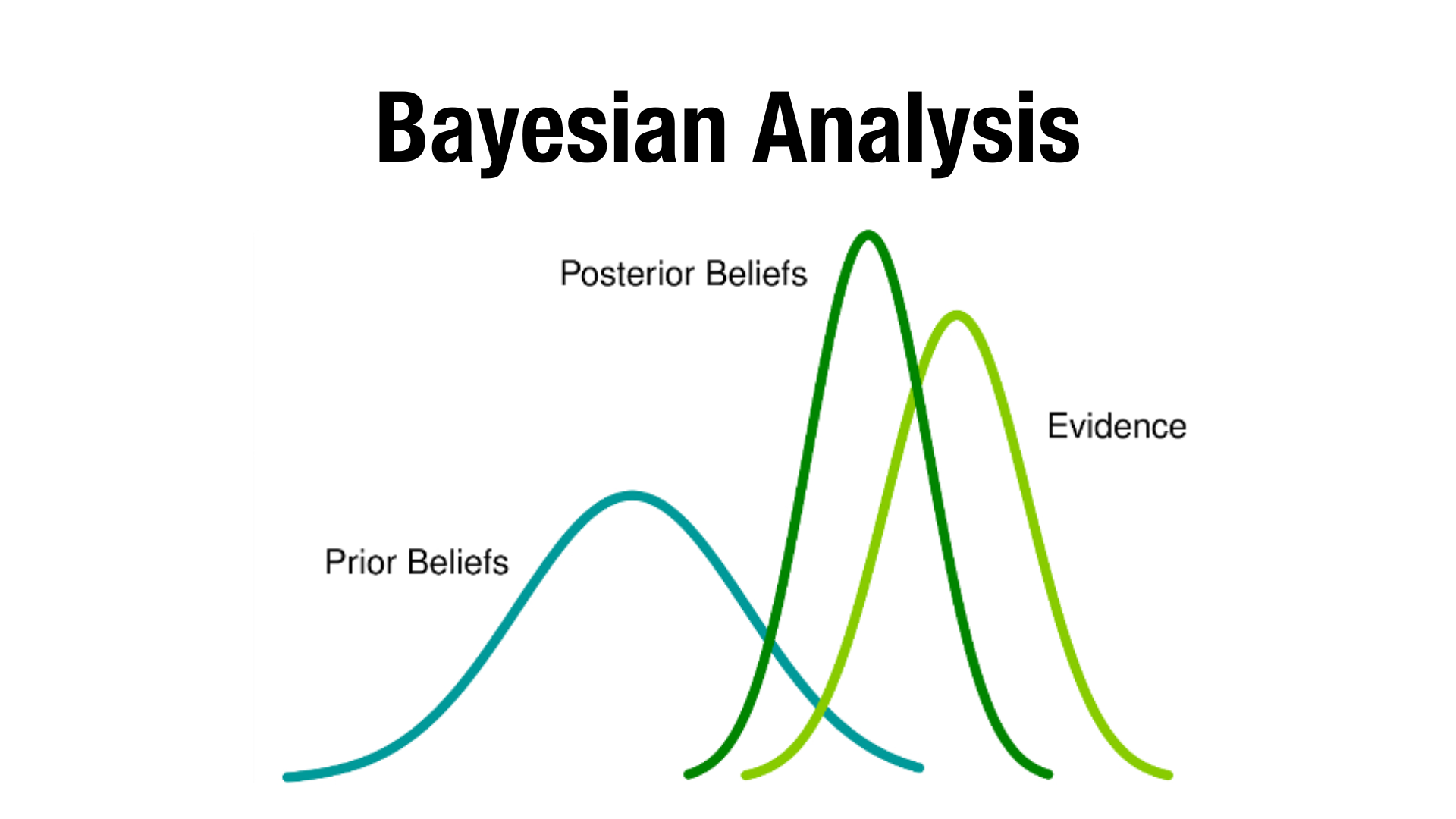
The approach works through three components:
1. Prior probability ;Your initial belief about something (say, how likely a drug is to work) based on existing knowledge like preclinical
data, earlier trials, or scientific understanding.
2. Evidence ; New data collected through your current trial.
3. Posterior probability ; Your updated belief that accounts for both prior knowledge and new data.
This contrasts sharply with traditional frequentist statistics, which has dominated clinical trials for decades. The frequentist approach treats each trial as an isolated experiment, fixes the sample size upfront, and asks a narrow question: If this drug doesn't work, how likely are these results by chance? Bayesian statistics flips the question entirely. Instead of asking about the probability of the data, it asks: Given everything we've observed, what's the probability this drug actually works? This produces a direct, intuitive answer"there's an 85% probability this treatment is effective" rather than the convoluted frequentist statement about hypothetical repetitions of the experiment.
Why This Matters for Clinical Trials
Bayesian methods enable adaptive trial designs where sample sizes can change, ineffective treatment arms can be dropped, and optimal
doses can be identified all while the trial is still running. This flexibility translates directly into efficiency. In the SHINE stroke trial, a Bayesian adaptive design reached the same conclusion as a traditional design but did so with 136 fewer patients and three months earlier. FDA Commissioner Marty Makary captured the regulatory shift in January 2026: "Bayesian methodologies help address two of the biggest problems of drug development: high costs and long timelines. Providing clarity around modern statistical methods will help sponsors bring more cures and meaningful treatments to patients faster and more affordably."
Digital Twins Create Virtual Patients That Reduce Trial Burden
Digital twins in the pharmaceutical context are AI-generated virtual representations of patients that predict future health outcomes, disease progression, and treatment responses. Unlike traditional modeling, which relies on simplified mechanistic assumptions, digital twins use machine learning trained on extensive real-world patient data to generate individualized, longitudinal forecasts.
The core application is straightforward: if you can accurately predict how a patient would do on placebo or standard care based on their
baseline characteristics, you need fewer actual control patients. Unlearn.AI, the leading company in this space, has developed Digital Twin Generators trained on data from over one million patients across 30+ disease areas. Their PROCOVA methodology which received regulatory qualification from the European Medicines Agency in 2022 can reduce control arm sizes by 25-50% while maintaining statistical rigor.
The business math is compelling. Consider a Phase 3 Alzheimer's trial requiring 674 patients at $500,000 per patient. Using digital twins to reduce the sample to 400 patients would save $137 million on that single trial. Johnson & Johnson's collaboration with Unlearn demonstrated that digital twins could reduce control arm sizes by up to 33% in Phase 3 Alzheimer's trials translating to 4-5 months of enrollment time saved.
Medidata, owned by Dassault Systèmes, offers a complementary approach with its Synthetic Control Arm built from 11+ million anonymized patients across 36,000+ clinical trials. Roche used Medidata's synthetic control arm to support EU market access for Alecensa across 20 European countries without waiting for Phase 3 completion. The FDA approved Merck's Bavencio for metastatic Merkel cell carcinoma based on a single-arm trial with a synthetic comparator.
Operational Impacts: How These Methods Transform Day-to-Day Trial Execution
Beyond the statistical elegance, Bayesian methods and digital twins deliver tangible improvements across the operational machinery of clinical trials. For clinical operations leaders, these aren't abstract methodological debates they translate directly into faster site activation, reduced protocol amendments, smarter interim decisions, and dramatically lower enrollment burdens.
Patient Recruitment and Enrollment
Patient recruitment remains the single largest source of trial delays. An estimated 85% of all clinical trials experience delays, with 80% of those delays tied to slow enrollment. The financial stakes are enormous: delays can cost sponsors anywhere from $600,000 to $8 million per day when accounting for lost sales of potential drugs. Nearly one-third of activated sites fail to enroll a single patient, representing wasted startup costs of $20,000-$30,000 per site plus $1,500 monthly maintenance.
Digital twins attack this problem directly by reducing the number of patients needed. For a trial with 1,000 patients, reducing the control group by 25% through digital twin methodology can cut enrollment time by four to five months. A 50% reduction can save nearly a year from the overall trial timeline. This isn't just about enrolling fewer patients faster it's about fundamentally changing the enrollment equation. Sites that would have been borderline performers under traditional designs become viable contributors when enrollment targets drop by a third.
The recruitment benefits extend to trial diversity as well. FDA guidance now requires diversity action plans, yet a Phesi analysis found that 42% of US cancer trials include no African-American patients and 48% have no Hispanic American representation. Digital twins enable more precise site selection by modeling which locations will successfully enroll diverse patient populations—improving both enrollment speed and regulatory compliance simultaneously.
Protocol Amendments and Trial Design
Studies show that 57% of protocols undergo at least one amendment, leading to significant time and financial losses. Each amendment triggers a cascade of operational disruptions: updated site training, revised informed consent documents, potential re-consent of enrolled patients, and regulatory resubmissions across multiple jurisdictions.
Bayesian adaptive designs reduce amendment frequency by building flexibility into the original protocol. Rather than fixing every parameter upfront and hoping the assumptions hold, adaptive trials specify in advance how the design will respond to accumulating data. Dose levels can be adjusted, treatment arms can be dropped, and sample sizes can be re-estimated all without the operational disruption of a formal amendment. Platform trials using Bayesian methods utilize protocol appendices rather than amendments when adding new treatment arms maintaining operational continuity while expanding scientific scope.
The operational benefits compound over time. Traditional trials often require amendments because initial assumptions about effect sizes,
enrollment rates, or patient characteristics prove wrong. Bayesian methods formally incorporate uncertainty about these parameters from the start, making the trial design more robust to real-world variation.
Interim Monitoring and Real-Time Decision Making
Traditional trial monitoring follows rigid schedules: interim analyses occur at predetermined timepoints, and decisions to continue, modify, or stop the trial require clearing statistical hurdles designed decades ago. Data monitoring committees often find themselves in awkward positions the data suggest the trial is unlikely to succeed, but frequentist stopping rules don't quite trigger early termination
Bayesian predictive probability transforms interim decision-making. Rather than asking whether current results cross a statistical threshold, it asks the operationally relevant question: If we continue this trial to completion, what's the probability we'll achieve our objective? This predictive framework aligns statistical analysis with the actual decisions sponsors and monitoring committees need to make.
The I-SPY COVID trial demonstrated this in practice, using Bayesian predictive probability at interim monitoring to inform decisions about
continuing randomization to specific treatment arms. When the analysis showed low probability of success for the backbone-plus-cyclosporine arm, the data monitoring committee recommended stopping randomization—a decision grounded in prediction rather than arbitrary statistical boundaries.
For trials with time lags between enrollment and outcome observation, Bayesian methods are particularly valuable. If patients take 90 days to reach the primary endpoint, traditional interim analyses can only use data from patients who completed follow-up—potentially ignoring valuable information from recently enrolled patients. Bayesian approaches can incorporate partial data from enrolled patients who haven't yet reached their endpoints, enabling more informed interim decisions without waiting for complete follow-up.
Site Selection and Performance Management
Site selection represents one of the most consequential operational decisions in trial execution, yet it has traditionally relied heavily on
investigator relationships and historical experience. Digital twin technology enables a more data-driven approach by modeling expected
enrollment performance, patient diversity, and protocol feasibility across potential sites.
The precision matters because site costs typically consume 30% of clinical trial budgets, and site performance varies dramatically. A small fraction of sites enroll the majority of patients, while underperforming sites contribute significantly to delays. AI-driven site selection using digital twin insights can improve enrollment speed by 10-20% by identifying top-performing sites earlier and more accurately.
Beyond initial selection, digital twins enable ongoing site performance optimization. Real-time monitoring against predicted enrollment curves allows early identification of underperforming sites, enabling resource reallocation before timeline slippage becomes critical. This shifts site management from reactive problem-solving to proactive performance optimization.
Data Management and Trial Operations Infrastructure
The operational infrastructure for Bayesian adaptive trials differs from traditional fixed designs. Enrollment rates must be carefully managed relative to outcome observation periods—if enrollment outpaces data accumulation, adaptations become impossible because results aren't available before all patients are enrolled. This requires tighter coordination between clinical operations and data management teams.
However, this integration creates opportunities for operational efficiency. Bayesian interim analyses can incorporate partial data and auxiliary variables, reducing the operational burden of ensuring every data point is complete before analysis. Risk-based monitoring approaches align naturally with Bayesian frameworks, focusing monitoring resources on high-risk sites and data elements rather than uniform source document verification.
The computational infrastructure has matured substantially. Custom code was once required for Bayesian trial simulations, but commercial platforms now enable standard adaptive design features with regulatory-accepted operating characteristics. This reduces the specialized expertise barrier and enables broader adoption across clinical operations organizations.
Real Drugs Approved Using These Methods Prove the Concept
These aren't theoretical possibilities; they're producing approved drugs with substantial commercial success.
Trulicity (dulaglutide) by Eli Lilly, which generated $7.1 billion in 2023 revenue, emerged from a Bayesian adaptive Phase 2/3 trial. The trial explored seven doses instead of the original three planned, with bi-weekly adaptive analyses updating randomization. The algorithm ultimately selected the 0.75mg and 1.5mg doses that became the marketed versions doses that might never have been tested under a traditional fixed design.
REBYOTA, approved in November 2022 as the first FDA-approved fecal microbiota product, used a Bayesian hierarchical model as its primary analysis for FDA approval. When enrollment difficulties prevented a second Phase 3 study, FDA agreed to let Ferring Pharmaceuticals formally integrate Phase 2 data through Bayesian methods. The analysis showed a 99.1% posterior probability that REBYOTA was superior to placebo—a direct, interpretable conclusion.
The I-SPY 2 trial platform has become the archetype for Bayesian adaptive oncology research. Running continuously since 2010, it uses
Bayesian predictive probability to identify promising breast cancer treatments that "graduate" when they show high likelihood of Phase 3
success. Seven drugs have graduated from the platform, with four receiving FDA marketing approval. The design achieved similar results in a fraction of the time with fewer patients with a maximum of 100 patients per experimental arm compared to traditional Phase 2 requirements.
Sanofi has built digital twins across dozens of disease conditions and used virtual patient modeling to bring Xenpozyme for Niemann-Pick
disease to regulators without running a large pediatric study. Their approach creates what executives call "scientific memory" continuously updated virtual patient models that accumulate institutional knowledge.
The Business Case Delivers Quantifiable Returns
McKinsey's analysis of AI-powered clinical trial optimization suggests transformative economics. A 12-month reduction in clinical development timeline adds over $400 million in net present value across a company's portfolio. Gen AI-powered CSR drafting alone increases NPV by $15-30 million per asset. Looking ahead, AI agents could boost clinical development productivity by 35-45% within five years.
The market reflects these opportunities. The AI in clinical trials market is projected to grow from $4 billion in 2025 to $25.7 billion by 2030.
Unlearn raised $50 million in Series C funding in January 2024, with Mira Murati (OpenAI's CTO) serving on its board. The digital twin market for clinical trials is projected to reach $38.4 billion by 2032, growing at a 42% CAGR.
Strategic Implications for Life Science Companies
Companies that develop capabilities in Bayesian methods and digital twins will enjoy compounding advantages. Time-to-market advantages directly translate to longer exclusivity periods and accelerated revenue. Capital efficiency improves as trial failures are identified earlier and resources redirect to promising candidates. And rare disease programs that would have been economically infeasible become viable.
The adoption path is becoming clearer. For organizations beginning this journey, the FDA recommends early engagement through the Complex Innovative Trial Design (CID) Meeting Program, which offers sponsors up to two meetings to discuss innovative designs. Internal capabilities in advanced statistics and machine learning are essential—these methods require more sophisticated expertise than traditional approaches.
Near-term priorities should include implementing AI-powered clinical study report generation (40% time reduction demonstrated), deploying AI for site selection and enrollment optimization, and piloting digital twin covariate adjustment in lower-risk trials.
Medium-term investments should focus on building platform trial capabilities and establishing external control arm methodologies for rare
disease programs.
The competitive dynamics are straightforward: companies that master these approaches will develop drugs faster, cheaper, and with higher success rates than those that don't. The regulatory path is now clear, the technology is maturing, and the business case is quantified. The question for executives is whether to lead this transition or follow it.